Recent advances in field-cycling NMR now make it possible to probe photo-CIDNP across controlled magnetic environments—transforming a long-standing theoretical prediction into measurable data.
This note highlights how the integration of LC-photo-CIDNP with a shuttling platform bridges spin-chemistry concepts with experimental precision, revealing a structured and field-tunable landscape of optical hyperpolarization.

An independent research team from New York Structural Biology Center (NYSBC) has recently published a peer-reviewed study in the Journal of Magnetic Resonance (2025), demonstrating high-field relaxometry using the Field-Cycling Shuttle System by Field Cycling Technology (DOI: 10.1016/j.jmr.2025.107989).
Their experiments, conducted between 1 T and 700 MHz (16.44 T), achieved full steady-state operation with rapid, mechanically stable shuttling. Importantly, all relaxation data were obtained without post-acquisition modeling, reconstruction, or machine-learning correction.
The results revealed highly reproducible mono-exponential relaxation curves, confirming that under precise mechanical control, the magnetic-field cycling process produces direct, physically interpretable signal decays suitable for quantitative analysis.
This study provides third-party validation that signal integrity in field-cycling NMR can be realized through robust engineering design, not computational compensation.
By minimizing transient oscillation and ensuring true mechanical settle before acquisition, the system enables reliable high-field relaxometry without algorithmic post-processing.
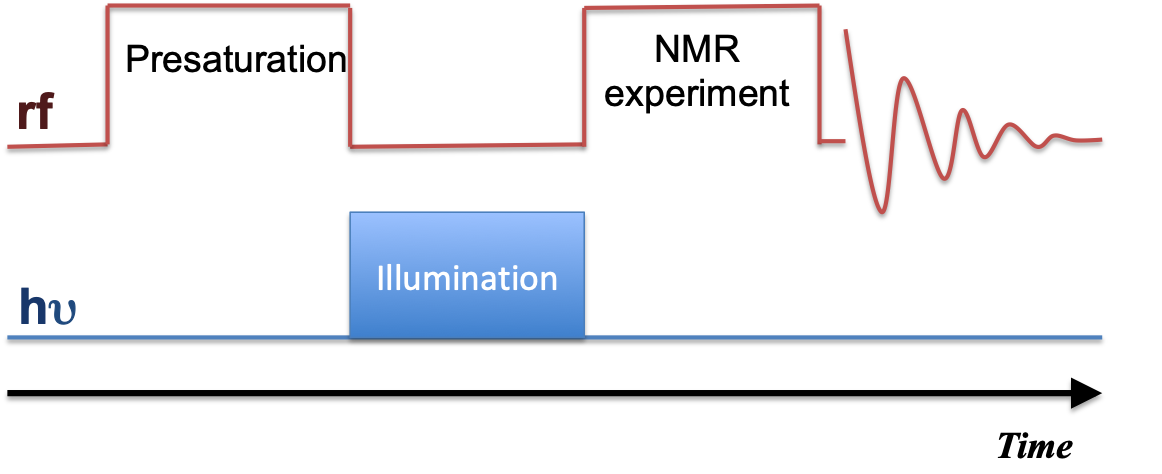
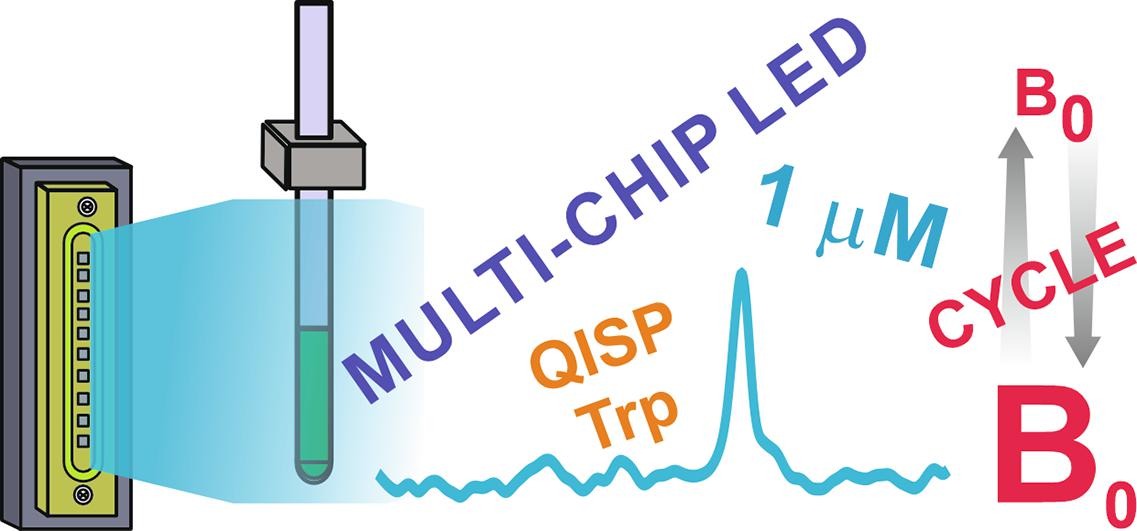
Illuminated Nuclear Magnetic Resonance (NMR) merges NMR spectroscopy with light irradiation, opening avenues in chemical analysis. This note highlights Photo-CIDNP (Chemically Induced Dynamic Nuclear Polarization) as a focal point, revealing its applications in radical reactions, and molecular dynamics, and uncovering hidden pathways.
The interplay between irradiation parameters and chemical reaction kinetics is pivotal. This note explored the correlation between total energy and reaction rates, emphasizing the role of pulse width in modulating reactions.
Light source selection, crucial in illuminated NMR, is dissected for lasers and Light-Emitting Diodes (LEDs). While lasers offer high irradiance and monochromaticity, LEDs provide broader emission and simple power control. In summary, illuminated-NMR leverages light-matter interactions to enhance sensitivity and probe dynamic processes. This article offers insights into applications, correlated techniques, and the transformative potential of illuminated NMR in diverse scientific fields.

This application note provides a comprehensive overview of hardware development for sample shuttling in high-field Nuclear Magnetic Resonance (NMR) spectroscopy. Sample shuttling involves the precise movement of NMR samples within the instrument using the stray field generated by the high-field NMR spectrometer. The technique facilitates investigations on molecular dynamics, interactions, and structural elucidation. The note emphasizes three crucial factors for hardware development: shuttling time, stability, and compatibility. Various sample shuttle systems from the literature are discussed in this note, highlighting their performance and contributions to this field. The comparison of sample shuttle systems recognized that work presented in T-h. Huang (2011) achieves the shortest shuttling time and highest compatibility with their mechanical field-cycling instrument. Other notable devices include those developed by S. Gross (1999), A. Redfield (2003), H. Stork (2008), F. Ferrage (2013), T. Theis (2020), and F. Ellerman (2023). These advancements provide researchers with options to enhance data collection and explore new avenues in high-field NMR spectroscopy.




