Field-dependent NMR relaxometry for Food Science
The investigation of molecular motions by Nuclear Magnetic Resonance (NMR) relaxometry is commonly conducted in material science and proton dynamics. Field-dependent NMR has already proven to distinguish different types of liquid samples, such as alcohols, or ionic liquids, etc. In many food applications, they mainly contain liquid substances. Different compositions and viscosities create significant patterns on NMR relaxation, R1, and R2, and diffusion coefficient D. Instead of the conventional approach by varying the temperature, the field-dependent measurement could avoid material changes by the thermal perturbation.
Due to the relationship of spin interaction and the correlation function, the Nuclear Magnetic Relaxation Dispersion (NMRD) can characterize several molecular motions in the observation systems. The information obtained from field-dependent relaxation is unavailable from conventional relaxometry, which is insensitive to slow processes.
In addition to the main fluctuations from internuclear interactions, there are magnetization transfer mechanisms that can contribute to relaxation. The chemical exchange of protons between water and accessible surface protons, for instance, amine, hydroxyl, and carboxyl groups, diffusion exchange dominated by physical movement of molecules, and the cross-relaxation, a through-space transfer of longitudinal magnetization between protons, such as protons on water near the surface of macromolecules and protons of macromolecule itself.
Foods are usually the mixtures of several ingredients and water. The interaction between molecules is not ignored. Thus, such chemical exchange or molecular interaction dominated the featured NMRD curves of various food applications.
In the following literature, they are demonstrating how NMRD profiles analyze the substances and relationships of those magnetization transfer mechanisms in food applications.
Related Literature
Field-dependent NMR relaxometry for Food Science: Applications and perspectives
Elif Gokcen Ates, Valentina Domenici, Małgorzata Florek-Wojciechowska, Anton Gradišek e, Danuta Kruk, Nadica Maltar-Strmecki, Mecit Oztop, Emin Burcin Ozvural, Anne-Laure Rollet
Trends in Food Science & Technology 110 (2021) 513–524
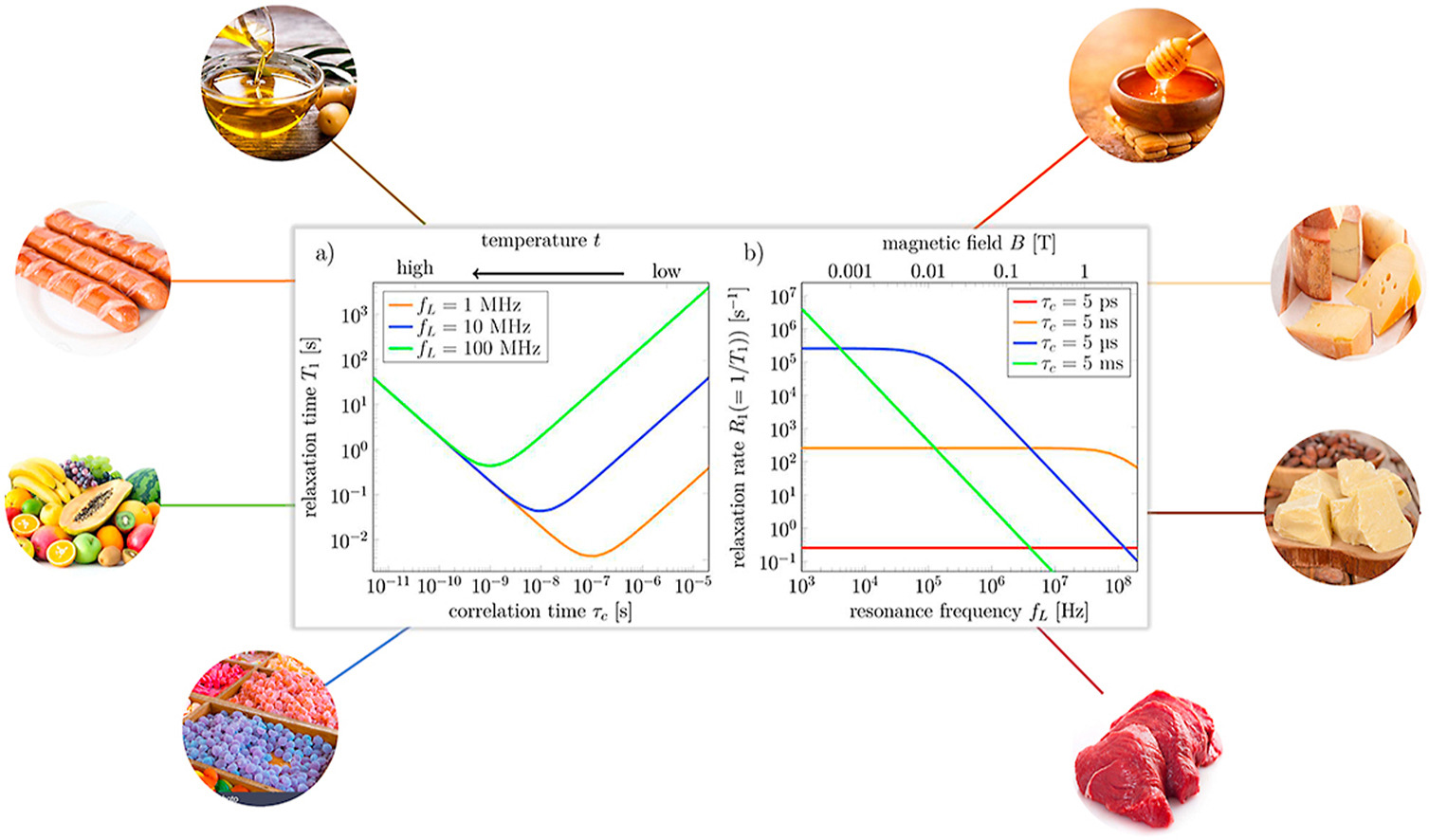
Background: Nuclear Magnetic Resonance (NMR) relaxometry is widely used for food products characterization. In order to exploit the full potential of the relaxation methods one can look at magnetic field dependence of spin-lattice relaxation. Analysis of relaxation dispersion characteristics of food systems may complement knowledge about composition and dynamics of their constituents, in particular, of water.
Scope and approach: In this article, we review the basic principles as well as recent applications of field-cycling NMR relaxometry in different food systems.
Key findings and conclusions: The inter- and intra-molecular interactions modulated by molecular dynamics can be efficiently studied with NMR relaxometry. This method can be exploited for discrimination of liquids, determination of diffusion coefficient or paramagnetic ion concentration, observation of crystallization/solidification processes. The technique has already proven to be helpful to investigate changes occurring in food products due to different processing techniques and to detect food adulteration. FFC benchtop instruments represent a new frontier in food studies.
NMR-Based Liquid Explosives Detector
Anton Gradišek, Tomaz Apih
Appl Magn Reson (2010) 38:485-493
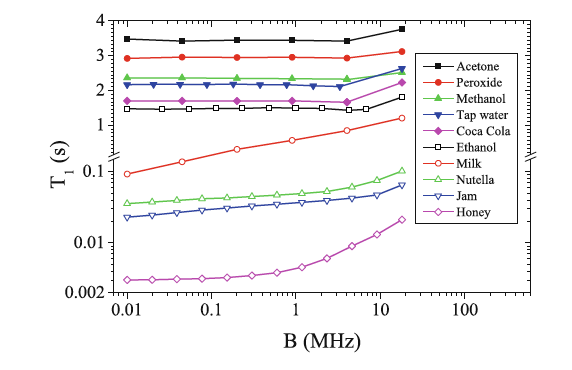
Liquid explosives pose a threat to security on airplanes and other public places, since they can easily be concealed as benign liquids. A detector, able to quickly identify liquids, would increase the possibility to detect such threats and speed up security checks. As a step towards a long-term goal to develop a liquid explosive detector, we have constructed an experimental setup based on a low-cost 1.1 T permanent magnet with huge static magnetic field gradient of 4.8 T/m, which allows us to measure proton relaxation times T1 and T2 and the self-diffusion coefficient D in liquid samples in a thin slice excited by radio-frequency pulses. We have developed a simple model in order to explain diffusion-enhanced nonexponential magnetization recovery in inversion recovery T1 experiment in this setup. Measuring a wide variety of liquid samples, we have demonstrated that it is possible to discriminate between the liquids based solely on these parameters. We discuss further improvements to the detection method, among those the choice of magnetic field, based on the fast field-cycling measurements.
Multiparameter NMR Identification of Liquid Substances
Andrey B. Konov, Kev M. Salikhov, Evgeniya L. Vavilova, and Bulat Z. Rameev
Magnetic Resonance Detection of Explosives and Illicit Materials, NATO Science for Peace and Security Series B: Physics and Biophysics
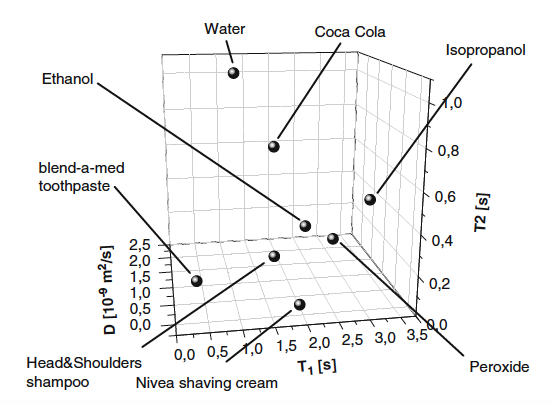
In this paper we study the method of distinguishing the substances by measuring their nuclear paramagnetic longitudinal and transverse relaxation times and the diffusion coefficient of molecules. Experiments performed using a commercial high magnetic field NMR spectrometer show the possibility to use this method for reliable identification of liquids. Observables in these experiments rather often cannot be described by a single exponential function. In the article we discuss how to utilize the non-single exponential experimental dependences for a quantitative processing of the NMR experimental results.